CD36-FITC, FA6.152, 100 Tests, CE
Product No:B49201
Product Specifications
| Target Species | Human |
| Specificity | CD36 |
| Other Names | CD36, GPIIIb, gpIV, PAS IV, platelet GPIV |
| Fluorochrome | FITC |
| Regulatory Status | CE-IVD |
| Size | 100 Tests |
| Format | Liquid |
| Flow Product Line | IOTest |
| Clone | FA6.152 |
| Isotype | IgG1 Mouse |
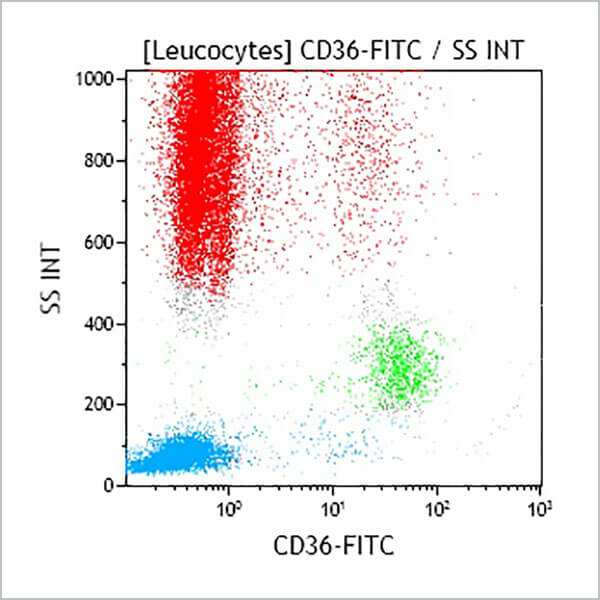
| Clone Description | The FA6-152 monoclonal antibody, raised against fetal erythrocytes, recognizes the CD36 family of antigens on platelets and certain hematopoietic cells. It does not react with lymphocytes or granulocytes. It reacts with both fetal and adult monocytes, megakaryocytes, platelets, and with reticulocytes. |
| Volume | 1 mL |